Abstract
Background:
Cancer screening is performed worldwide for several malignancies. Monoclonal gammopathy of undetermined significance (MGUS) precedes multiple myeloma (MM) and related lymphoproliferative disorders (LP). However, less than 5% of all MM patients are diagnosed during their precursor state and individuals who develop MM while being monitored for MGUS have better overall survival and fewer complications, compared to MM patients diagnosed without knowledge of MGUS. Thus, population-based screening for MGUS could identify candidates for early treatment of MM/LPs. To evaluate whether systematic screening is beneficial, we performed the Iceland Screens, Treats, or Prevents Multiple Myeloma (iStopMM) study, the first population-based screening study for MGUS that includes a randomized clinical trial (RCT) of follow-up and treatment strategies.
Methods:
All living residents of Iceland on September 9th, 2016 who were born before 1976 (N=148,708) were invited to participate. Of those, 80,759 (54.3%) provided informed consent for screening. Serum samples were collected from participants alongside clinical blood sampling in the Icelandic health service between September 2016 and the end of 2020. All samples were shipped to the Binding Site in Birmingham, UK, for screening. Samples were tested for M-proteins by capillary zone electrophoresis and immunofixation electrophoresis performed to confirm and characterize suspected M-proteins. Free light chains (FLCs) were measured using the FreeLite® assay. Individuals with a previous diagnosis of MM/LPs/MGUS (N=237) were excluded. Per protocol and informed consent, participants with MGUS were randomized to one of the three study arms: Arm 1 where participants are not contacted; Arm 2 where individuals are followed based on current guidelines; and Arm 3 where individuals are followed with a more intensive diagnostic and monitoring strategy. Participants who progress are offered early treatment. All participants repeatedly answered questionnaires on quality of life and mental health.
Results:
A total of 75,422 participants (93.4%) provided a serum sample for screening. Of those, 3,725 (4.9%) had MGUS. The prevalence of MGUS was dependent on age with 2.3%, 6.2%, and 12.9% diagnosed in age groups 40-59, 60-79, and 80-103 years, respectively. The prevalence of MGUS was higher in males, 5.9% vs 4.1% (p<0.0001). Most individuals with MGUS had either low-risk (38%) or low-intermediate (36%) risk MGUS, followed by high-intermediate (26%) risk MGUS. High-risk MGUS was only present in 0.2% of MGUS cases (n=9).
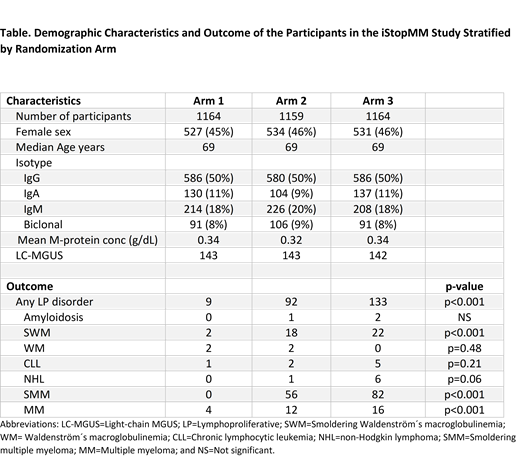
The RCT includes 3,487 newly diagnosed MGUS individuals with 1164, 1159, and 1164 individuals in arms 1, 2 and 3, respectively (Table). The median age at diagnosis was 69 years in arms 1 and 2, and 70 years in arm 3. Females constituted 45.9% and the isotypes were IgG (50%), IgA (10%), IgM (18%) and biclonal (8%). The median M-protein concentration was 0.34 g/dL. A total of 428 light-chain MGUS cases were randomized. The demographic distribution was well balanced between the three arms.
After a median follow-up of 3 years, 194 patients in the RCT have been diagnosed with any LP: 9 in arm 1, 92 in arm 2, and 133 in arm 3 (p<0.001). The participants in arm 1 were diagnosed with smoldering Waldenström's macroglobulinemia (SWM)(N=2), WM (N=2), chronic lymphocytic leukemia (CLL) (N=1), and MM (N=4). Participants in arm 2 were diagnosed with amyloidosis (N=1), SWM (N=18), WM (N=2), CLL (N=2), non-Hodgkin lymphoma (NHL) (N=1), smoldering MM (SMM) (N=56), and MM (N=12). Participants in arm 3 were diagnosed with amyloidosis (N=2), SWM (N=22), CLL (N=5), NHL (N=6), SMM (N=82), and MM (N=16). The difference between study arms was statistically significant for all LPs combined, and for SWM, SMM, and MM (Table).
Conclusion:
In this large prospective population-based screening study including >75,000 screened persons, we have identified 3,725 individuals with monoclonal gammopathy. In the RCT, after 3 years of follow-up, we show that active screening identifies significantly higher number of individuals with full-blown malignancy and smoldering disease, illustrating the fact that early detection and intervention is achievable. Although our findings are encouraging, until final results of the iStopMM study become available, including data on survival and quality of life, we advise against systematic MGUS screening in healthy individuals.
Kristinsson: Amgen: Research Funding; Celgene: Research Funding. Kampanis: The Binding Site: Current Employment. Hultcrantz: Curio Science LLC: Consultancy; Daiichi Sankyo: Research Funding; GlaxoSmithKline: Membership on an entity's Board of Directors or advisory committees, Research Funding; Amgen: Research Funding; Intellisphere LLC: Consultancy. Durie: Amgen: Other: fees from non-CME/CE services ; Amgen, Celgene/Bristol-Myers Squibb, Janssen, and Takeda: Consultancy. Harding: The Binding Site: Current Employment, Membership on an entity's Board of Directors or advisory committees, Patents & Royalties. Landgren: Janssen: Research Funding; Celgene: Research Funding; Janssen: Honoraria; Janssen: Other: IDMC; Amgen: Honoraria; Takeda: Other: IDMC; Amgen: Research Funding; GSK: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal